 As of the end of last month, Bard faced 886 lawsuits. Such is the backdrop for the growing number of Bard IVC filter lawsuits. The report even got the attention of lawmakers, like Senator Charles Grassley (R-IA), who last year sent a letter to the FDA, inquiring about the agency’s oversight of Bard filters. The problems with Bard IVC filters were broadcast throughout the country when NBC News ran a pointed investigative report on the company. The company continued to sell the G2 model until 2010. According to the G2 model, however, did not resolve any of the issues associated with the Recovery model. Rather than issue a recall, Bard replaced the Recovery model in 2005 with the G2 model. Shortly thereafter, the FDA began receiving adverse event reports of death and injury, including migration and breakage, among patients who received a Recovery filter. Bard received approval from the FDA in 2002 to sell its Recovery IVC filter. Bard IVC Lawsuits Allege Company Knew of Health Risks for YearsĬ.R. Migration and breakage are at the heart of both the Bard and Cook IVC filter lawsuits. Pieces of IVC filters have also been known to break and migrate elsewhere in the body, occasionally ending up in arterial tissue or other vital internal organs. The problem with IVC filters is that they occasionally migrate from their primary insertion point toward the lungs or the heart. But studies have shown that the vast majority of IVC filters are never successfully retrieved, which only increases the risk of serious side effects. Food and Drug Administration (FDA) has recommended that IVC filters be removed 29-54 days following initial implantation. Most IVC filters are designed as a temporary measure and are to be retrieved once the imminent danger of blood clot migration has passed. 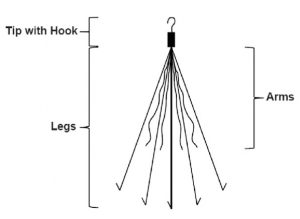
IVC filters are intended for patients who are at high risk for developing blood clots, and for whom blood-thinning medication is either inappropriate or ineffective. Their purpose is to capture blood clots that move from the lower body toward the lungs or heart. IVC filters are medical devices that are surgically implanted in the inferior vena cava (IVC), a large vein that transports deoxygenated blood from the lower body to the heart. Bard, which is particularly astounding because Bard filters have been the subject of scrutiny for over a decade. Today, more people have filed lawsuits against Cook Medical than C.R. Back then, the number of Cook IVC filter lawsuits was dwarfed by suits against one of Cook’s largest competitors, C.R. People harmed by IVC filters made by Cook Medical have been filing lawsuits against the medical device manufacturer since 2012. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
